Following alarming reports of kidney failure and multiple child fatalities, the Government of India has issued an urgent advisory cautioning against the prescription of cough syrups for children under two years of age. The directive calls on state governments, health authorities, and medical practitioners to exercise strict restraint and follow evidence-based protocols when treating pediatric respiratory symptoms.
The Trigger: Child Deaths and Health Crisis
The advisory comes in the wake of reports from districts in Madhya Pradesh where several children suffered acute kidney injury after being administered cough syrups. Health officials believe that contaminated or improperly formulated syrups may have contributed to these tragic outcomes. Given the severity and pattern of the cases, central health agencies have escalated the matter, prompting a nationwide response.
Investigations are underway to determine whether toxic impurities, off-label combinations, or dosage errors are involved in these incidents. Meanwhile, states have been asked to review pediatric prescription patterns and trace the circulation of suspect syrup batches.

Key Directive in the Advisory
The central government’s notice emphasizes rational and evidence-based use of cough syrups in children. It specifically advises that doctors should not prescribe cough syrups to infants below two years, except under exceptional, documented circumstances where the benefits clearly outweigh risks.
Health departments are instructed to:
- Strictly monitor pediatric prescription practices.
- Withdraw availability of suspect syrup batches pending safety verification.
- Enhance pharmacovigilance, including adverse event reporting and regulatory audits.
- Issue local circulars reinforcing the advisory to all doctors, particularly in rural and primary health care settings.
- Enforce regulatory oversight on manufacturing, inspection, and supply chains of pediatric formulations.
Broader Background and Precedents
Indian authorities have previously flagged certain fixed-dose cough syrup combinations, especially those combining decongestants, antihistamines, or expectorants, as problematic in young children. In some regions, health authorities have already suspended or tightened protocols around syrups containing dextromethorphan and related agents for children under four years.
These new fatalities echo prior global episodes—such as children suffering from kidney failure after ingesting contaminated syrups in other countries—underscoring the need for precaution and stronger quality control.
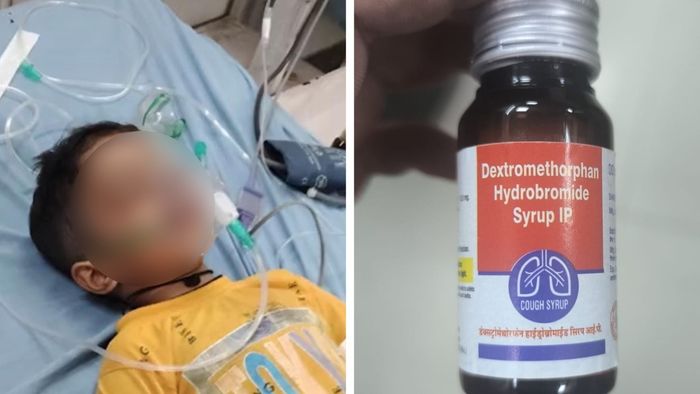
Clinical and Policy Implications
For pediatricians and medical practitioners, the advisory challenges routine cough syrup prescriptions. Physicians will need to rely more on nonmedication approaches — hydration, humidified air, saline nasal washes, rest — especially in infants with viral coughs, which tend to resolve on their own. When medication is essential, any choice must be backed by safety data and supported by careful monitoring.
Policywise, the advisory may spur deeper reforms in drug regulation, batch testing, and oversight of over-the-counter pediatric medications. It also raises critical questions about pharmaceutical accountability, public health safeguards, and how vulnerable populations depend on preventive systems.
What to Watch Next
- The findings of central and state-level investigations into syrup toxicity and causation.
- Official lists of suspicious or banned syrup batches and the scope of recalls.
- Compliance from state health departments and district medical authorities in implementing the advisory.
- Possible legal or regulatory repercussions against manufacturers or distributors found culpable.
- Public communication campaigns to reassure parents and guide care for young children’s cough symptoms.
Amid public grief and outrage, the government’s move signals urgency. But its effectiveness will depend on decisive follow-through, transparent inquiry, and safe, trusted alternatives for children’s respiratory care.


Leave a Reply